Plastic Bottles (HDPE, LDPE, PET, PP), Moulded Glass Bottles and Vials (Type I, II, III), Tubular Glass Ampoules / Vials / Cartridges, Blister Foil, Pouches, Strip-Packs, Sachets, Aluminium and Plastic Tubes, Stoppers, Caps & Closures, Pre-Filled-Syringes, COC Vials, Microneedle Patches, Metered Dose Pumps, Inhalers, Nebulizers, Drug-Device Combinations, Auto-Injectors, Pens, Self-Injection-Devices, Wearables
Primary pharmaceutical packaging comes in direct contact with the medicine or drug. They generally include plastic bottles, glass bottles, ready-to-fill syringes, pre-filled inhalers, medication tubes, blister packs, and others. It’s a broad range of different packaging materials and combinations and also devices used for the various types of drug delivery and routes of administration.
Routes of Administration define the type and construction of the primary packaging and devices. Major routes of administering medications are e.g., Oral, Parenteral, Topical, Transdermal, Intranasal, Inhaled, Ocular/Ophthalmic.
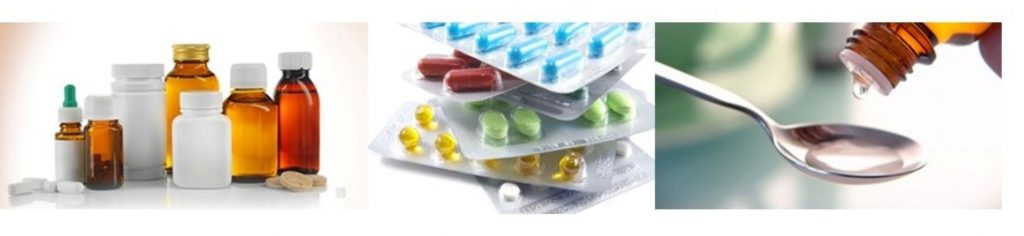
Oral Drug Delivery

Solid and liquid oral drugs like e.g., tablets, capsules and syrups— the pharma industry’s most established dosage forms — are cost-effective to manufacture, shelf stable and easy to administer. Despite the rise of biologics, oral drugs continue to play a pivotal role in the industry’s quest to produce safe, effective treatments.
Based on drug delivery, the oral drug delivery mode is the largest category in the overall pharmaceutical packaging market.
The specific needs of the elderly and handicapped are being integrated more and more into packaging design, thus making the product more convenient to use. Brand protection to prevent counterfeiting is also a significant factor that enhances competition in the market. The development of new packaging types such as child-resistant packaging (CRP), and others with improved properties have led to the rapid growth of the market.
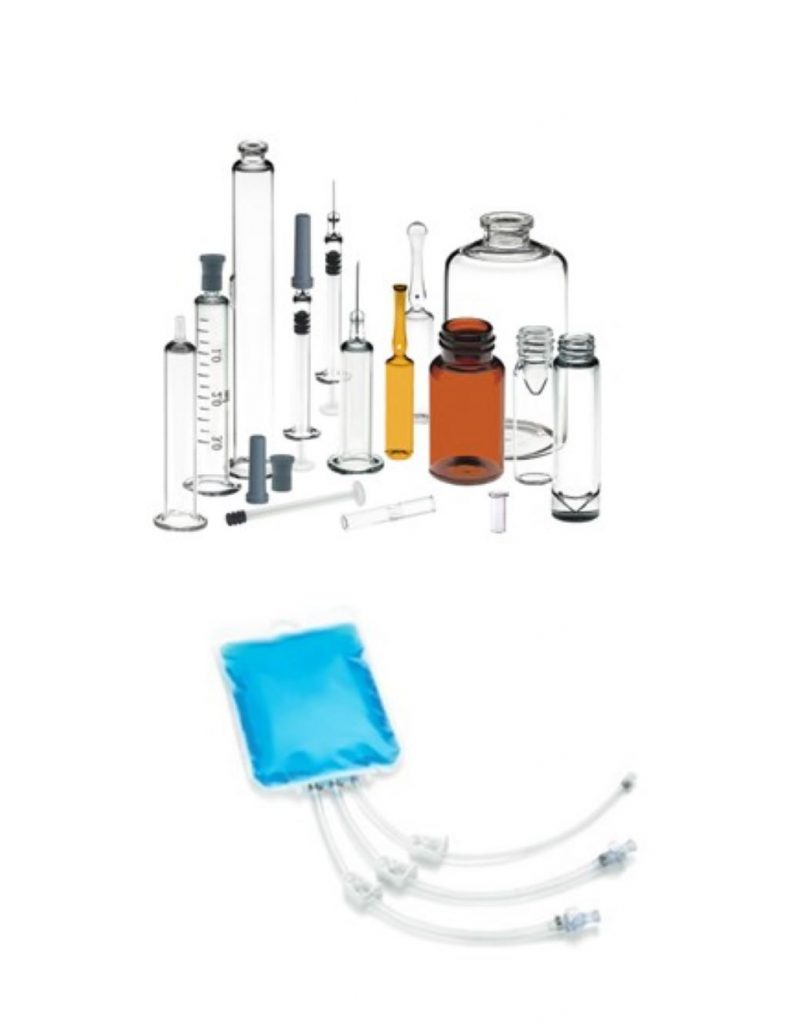
Parenteral Drug Delivery
Advancements in biotechnology, new oncologic drugs and the revolutionizing treatment options of Cell & Gene therapies are just some few examples for growth drivers in the market for injectables and infusion and its sterile fill-finish processes. Viral diseases like Covid did push the market for vaccines filled in tubular vials dramatically.
The range of dosage forms to meet the large molecule’s needs comprises e.g.
- Small and large volume parenterals in glass vial/bottles and plastic
- Lyophilized vials
- Pre-filled syringes and cartridges, pens and auto-injectors
The long-term shift in the parenteral market and drug developments, coupled with the demand for more patient centric delivery formats such as prefilled syringes and autoinjectors, is driving the growth for the sterile fill-finish market.

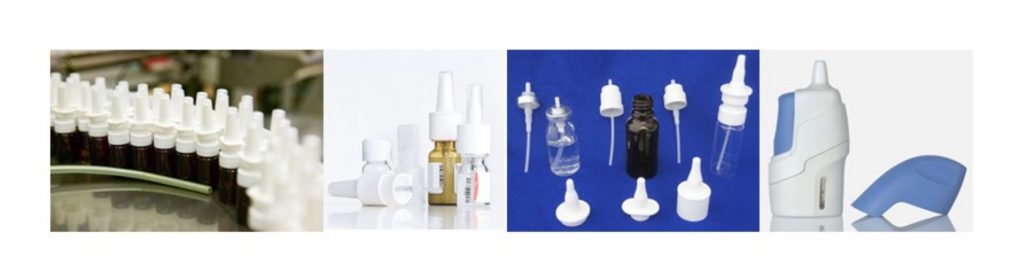
Inhaled Drug Delivery

The growth of the inhaler segment is expected to be supported by the rising number of chronic asthma and allergy, treated with inhalation drugs. Inhalers are the major product segments.
Dry-powder inhalers (DPIs), Nebulizers, and pressurized metered-dose inhalers (pMDI) are the most common drug delivery devices in this segment.
Even large molecules such as peptides, antibodies, and various types of engineered proteins can now be delivered via the lungs. Inhalation provides an alternative to parenteral delivery of these active substances.
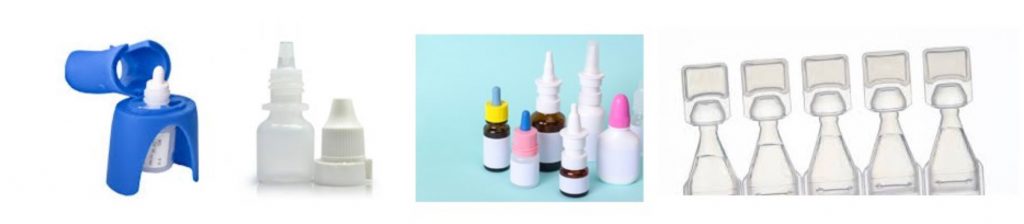
Intranasal Drug Delivery
The intranasal delivery route is frequently used for the local treatment of allergic rhinitis, nasal congestion, and cold symptoms. However, it has emerged as an alternative route of systemic delivery for a variety of indications such as migraine, seizures, breakthrough pain, and opioid overdose amongst others.
The nasal route is particularly suitable for the delivery of drugs that require a rapid onset of action. It has advantages over both oral and parenteral administration.
In recent years, there has been increased interest in the intranasal delivery route. The majority of the market (units) are OTC products (Over-the-Counter), filled in HDPE (high-density polyethylene) bottles and fitted with dosing pumps and protective caps.
Ocular Drug Delivery
In the past two decades, the arena of ocular drug-delivery technologies has dynamically advanced and resulted in newer therapeutic interventions for chronic ocular disorders. Topical delivery of ophthalmic formulations is the most preferred route for the delivery of therapeutic agents to the anterior segment of the eye.
More than 70% of ophthalmic drug products are simple solutions supplied in multi-dose plastic container closure systems (CCS).
Dermal and Transdermal Drug Delivery
Where applicable, therapy options that are available in a topical format are often preferable for patients, compared to alternatives that may involve injection or oral dosing. This preference is an attributable factor to the growth in the topical pharmaceuticals market.
Selecting the most appropriate packaging option for today’s topical formulation route brings its own challenges for cremes, ointments and gels. The goal is to balance the need for proper dose dispensing or metering while safeguarding the product against degradation from exposure to light or oxygen.




